更多“K£102A润滑油压力一级报警值为0.25MPa,当油压低…”相关的问题
更多“K£102A润滑油压力一级报警值为0.25MPa,当油压低…”相关的问题
第6题
(I.Johanhen模型)设K=K(t),H=H(t)分别为某国t时刻的资本存量、外援水平,它们满足如下方程:K'=aK+H,H'=BH其中a,β为正的常数.已知K(0)=K0</sub>>0,H(0)=H0</sub>>0.求K(t),H(t).
第7题
反应A→B,且CB.O=0,CA.O≠0.若反应物A完全转化为B所需时间为t并测得t/t1/2=2,则此反应的级数为().
反应A→B,且CB.O=0,CA.O≠0.若反应物A完全转化为B所需时间为t并测得t/t1/2=2,则此反应的级数为().
点击查看答案
A.零级
B.一级
C.3/2级
D.二级
第8题
100℃时下列反应的Kθ=8.1×10-9,=125.6J·mol·K-1.计算:(1)100℃,总压为200KPa
100℃时下列反应的Kθ=8.1×10-9,=125.6J·mol·K-1.计算:(1)100℃,总压为200KPa
点击查看答案
100℃时下列反应的Kθ=8.1×10-9,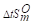 =125.6J·mol·K-1.计算:
=125.6J·mol·K-1.计算:
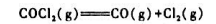
(1)100℃,总压为200KPa时COCl2的解离度;
(2)100℃下上述反应的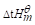 ;
;
(3)总压为200kPa,COCl2的解离度为0.1%时的温度,设ΔTGp,m=0.
第10题
为了燃机的安全运行,当燃机排气压力高报警时,燃机()。但当燃机排气压力高到跳闸值时,燃机保护系统发出()。
为了燃机的安全运行,当燃机排气压力高报警时,燃机()。但当燃机排气压力高到跳闸值时,燃机保护系统发出()。
点击查看答案
第11题
A sample of D-ribose(C5H10O5) with mass 0. 727 g was weighed into a calor
A sample of D-ribose(C5H10O5) with mass 0. 727 g was weighed into a calor
点击查看答案
A sample of D-ribose(C5H10O5) with mass 0. 727 g was weighed into a calorimeter and then ignited in presence of excess oxygen. The temperature rose by 0.910 K when the sample was combusted. In another separated experiment in the same calorimeter the combustion of 0. 825 g of benzoic acid,for which the ΔcUΘm= 3251 kJ·mol-1,gave a temperature rise of 1.940 K. Calculate the ΔrUΘmand ΔrHΘmof D-ribose combusted.


 如果结果不匹配,请
如果结果不匹配,请