更多“水中CaO含量在80mg·L-1以下称之为软水。()”相关的问题
更多“水中CaO含量在80mg·L-1以下称之为软水。()”相关的问题
第1题
为了确保水泥熟料的安定性,应控制f£CaO的含量,一般回转窑熟料控制在()以下,立窑熟料控制在()以下。
为了确保水泥熟料的安定性,应控制f£CaO的含量,一般回转窑熟料控制在()以下,立窑熟料控制在()以下。
点击查看答案
第3题
A 0. 2236 g sample of lime stone was dissolved in dilute HCL. After .(NH4)2C
A 0. 2236 g sample of lime stone was dissolved in dilute HCL. After .(NH4)2C
点击查看答案
2
O4was introduced and the pH of the resulting solution was adjusted to permit the quantitative precipitation of CaC2O4, the solid was isolated by filtration, washed free excess C2O42-,and dissolved in dilute H2SO4. Titration of the liberated H2C2O4, required 26.77mLof 0.02356 mol·L-1KMnO4. Calculation the mass fraction of CaO in the sample.
第6题
称取石灰石试样0.2503g,溶于盐酸制备成溶液后,移入100mL容量瓶中加水稀释至刻度。吸取25.00mL该
试样,在pH=12时,以KB为指示剂,用浓度为0.02500molL1的EDTA标准溶液滴定,消耗24.00mL。计算试样中以CaO表示的含量。
点击查看答案
(已知CaO的摩尔质量58.06gmol1)
第7题
假如有一邻苯二甲酸氢钾试样,其中邻苯二甲酸氢钾含量约为90%,余为不与碱作用的杂质,今用酸碱滴
定法测定其含量.若采用浓度为1.000mo1·L-1的NaOH标准溶液滴定之,欲控制滴定时碱溶液体积在25mL左右,则:
点击查看答案
(1)需称取上述试样多少克?
(2)以浓度为0.0100mol·L-1的碱溶液代替1.000mol·L-1的碱溶液滴定,重复上述计算.
(3)通过上述(1)(2)计算结果,说明为什么在滴定分析中常采用的滴定剂浓度为0.1-0.2mol·L-1.
第8题
水硬度的单位是以CaO为基准物质确定的,10为1L水中含有()A.1gCaOB.0.1gCaOC.0.01gCaOD.0.001gCaO
水硬度的单位是以CaO为基准物质确定的,10为1L水中含有()
A.1gCaO
B.0.1gCaO
C.0.01gCaO
D.0.001gCaO
第9题
25℃,,CO2饱和溶液的浓度为1.5×10-5mol·L-1,且假定溶于水中的CO2均生成了H
25℃,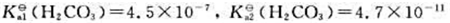 ,CO2饱和溶液的浓度为1.5×10-5mol·L-1,且假定溶于水中的CO2均生成了H2CO3试通过计算说明酸雨的pH应该小于多少?
,CO2饱和溶液的浓度为1.5×10-5mol·L-1,且假定溶于水中的CO2均生成了H2CO3试通过计算说明酸雨的pH应该小于多少?


 如果结果不匹配,请
如果结果不匹配,请