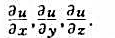题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
[单选题]
X(g)+3Y(g)= 2Z(g) △H=-92kJ/mol,将1mol X和3mol Y通入2L的恒容密闭容器中进行反应,10min时测得Y的物质的量为2.4mol。下列说法正确的是()
A.10min内,X的平均反应速率为0.01mol/(L·min)
B.第10min时,X的反应速率为0.01mol/(L·min)
C.10min内,消耗0.1mol X,生成0.4mol Z
D.10min内,X和Y反应放出的热量为92kJ
 答案
答案
A、10min内,X的平均反应速率为0.01mol/(L·min)
 如果结果不匹配,请 联系老师 获取答案
如果结果不匹配,请 联系老师 获取答案

 更多“X(g)+3Y(g)= 2Z(g) △H=-92kJ/mol…”相关的问题
更多“X(g)+3Y(g)= 2Z(g) △H=-92kJ/mol…”相关的问题